Sometime in the next two months the Food and Drug Administration is expected to decide whether to give the go-ahead for an implant designed to treat addiction to heroin or prescription painkillers. Known as probuphine, the implant releases medication that helps prevent withdrawl and reduces cravings over the course of six months. An FDA advisory committee voted 12-5 to recommend its approval. But the product has raised some safety concerns
The development of probuphine comes at a time when the White House, along with health organizations, are recommending the expanded use of medications to treat opiate addiction. The most effective medications are methadone and buprenorphine, commonly known as Suboxone. Both cut the risk of death from overdose in half and substantially reduce infections from IV drug use such as HIV and Hepatitis C, according to the Substance Abuse and Mental Health Services Administration.
Probuphine, says Behshad Sheldon, president and CEO of Braeburn Pharmaceuticals, delivers a steady dose of buprenorphine through four matchstick-size implants in a patient’s arm.
“I think long-acting products are a game changer in almost every therapeutic area," she says. "I think they're absolutely a game changer when it comes to opioid use disorder. I am confident probuphine itself is going to help alot of patients. We've had patients say that it changes their lives.”
Probuphine is specifically intended for patients who have been taking buprenorphine successfully for awhile. And Sheldon, whose company is manufacturing the implant, says it has several advantages over buprenorphine pills or films that patients dissolve under the tongue.
No. 1 is that it helps patients stay compliant with treatment. They don’t have to worry about forgetting to take the medication.
“There’s also no possibility of losing your medication or having it stolen or having it accidentally taken by children,” Sheldon says. “The medication doesn’t end up on the street.”
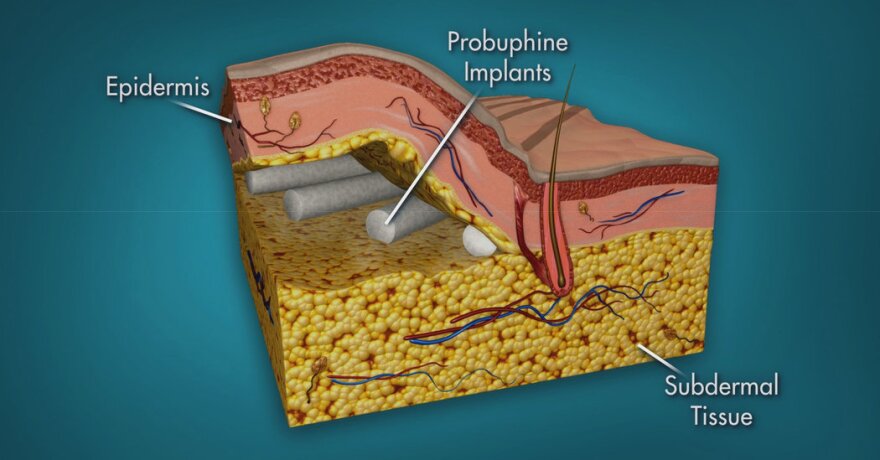
Diversion has been an unintended consequence of medication-assisted treatment, as some addicts buy the drugs on the street it in an effort to stave off withdrawal, and others sell their medication to buy other drugs.
Despite these potential advantages of a long-acting maintenance medication for addiction, there are concerns about the safety of probuphine. Addiction specialist, Dr. Mark Publicker, who has a Portland-based Suboxone practice, says he has concerns about what might happen to someone with the implant in an emergency where surgery or anaesthesia becomes necessary.
“Just in terms of — would somebody treating a person in an emergency room actually know that the person had this device or know how to remove it?” he says.
Dr. Publicker says that knowledge can distort study results since there’s a real desire to demonstrate the effectiveness of the drug. He also has concerns about what might happen to someone with the implant in an emergency where surgery or anesthesia becomes necessary.
“Just in terms of, would somebody treating a person in an emergency room actually know that the person had this device or know how to remove it?” he says.
Insertion of probuphine under the skin takes about 15 minutes. Removal takes a little longer. And if it's approved by the FDA doctors will have to be trained on how to do both.
What concerns pharmacist Tracy Rupp is the transition of buprenorphine patients to the implant. Dr. Rupp is with the National Center for Health Research which analyzes health information. She says some patients on the implant needed to take additional doses of buprenorphine and she'd like to see further study about the risks and benefits for patients who make the switch.
“Patients in he implant group actually required a lot more extra oral doses of medication than what the patients who were just given the oral doses to begin with and so the question is, well what patients is this really appropriate for if they're still going to need a whole lot of oral doses in addition to the implant?” she says.
But at a public hearing on probuphine in January, patients, doctors and clinical trial investigators pointed to the same clinical study that showed at THE end of six months, 85 percent of patients given probuphine showed no evidence of illicit drug use compared to 72% of patients on the oral form of buprenorphine and they urged the FDA to approve it.
***Editor's Note: In an earlier version of this story Dr. Mark Publicker was quoted as saying that he was concerned that the clinical trial was an "open label" study that could distort results. In fact the clinical trial was a randomized and double blind study. Dr. Tracy Rupp was also quoted as saying that it takes three or four weeks for the implant to reach therapeutic levels and leaves patients potentially at risk for relapse or overdose. She has clarified her statement that it takes three to four weeks for the therapeutic levels to stabilize, and that leaves patients potentially at risk for relapse or overdose during those weeks.



